 |
 |
- Search
| Obstet Gynecol Sci > Volume 66(5); 2023 > Article |
|
Abstract
Objective
We aimed to evaluate the usefulness of delta neutrophil index (DNI), a new inflammatory marker, in polycystic ovary syndrome (PCOS).
Methods
This retrospective case-control study was conducted at a tertiary health center. The DNI and other blood parameters obtained from the complete blood count examination of 227 individuals, consisting of 72 PCOS patients and 155 controls, were compared between the two groups. A receiver operating characteristic analysis was performed to examine the relationship between DNI and PCOS.
Results
DNI, white blood cell (WBC) count, and neutrophil count were significantly higher in the PCOS group than in the control group (P=0.028, 0.011, and 0.037; respectively). DNI and WBC counts were significantly higher in nonobese-PCOS patients (P=0.018 and 0.041; respectively). When the obese-PCOS and obese-control groups were compared, only neutrophil count was significantly higher in obese-PCOS patients (P=0.016). Significance was observed at cut-off values of 0.015 (area under the curve [AUC]=0.588) (P=0.034; sensitivity, 78%; specificity, 35%; Youden’s index=0.133) for DNI: 9.35 (AUC=0.594) (P=0.022) for WBC; and 5.38 (AUC=0.628) (P=0.002) for neutrophils.
Conclusion
Higher DNI in PCOS patients and similar results in the non-obese-PCOS group were observed when obese and non-obese-PCOS patients were considered separately. However, the lack of difference in the obese-PCOS group strengthens the hypothesis that there is obesity-independent inflammation in PCOS.
Polycystic ovary syndrome (PCOS) is one of the most common endocrinopathies in women of reproductive age, with a prevalence of 7-10% [1]. It is a complex, multifactorial metabolic disorder characterized by oligoanovulation, hyperandrogenism, and polycystic ovaries [2]. However, its specific causes are still unknown [3]. Some evidence indicates that low-grade inflammation is a potential risk factor for PCOS and plays an essential role in the development of insulin resistance, type 2 diabetes mellitus, and atherosclerosis [4]. In PCOS patients, proinflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-6, IL-8, and IL-18 [5]; C-reactive protein (CRP); and homocysteine levels are high [5,6]. The level of leptin, which induces interferon-gamma and IL-6 expression and mostly originates from the adipose tissue, also increases in PCOS patients [7].
Hatziagelaki et al. [8] found body mass index (BMI)-independent correlations among chemokines, sex steroids, and pituitary hormones in PCOS patients. These data revealed a crucial link between PCOS and chronic inflammation. However, analyzing these molecules is difficult, including calculation of the exact threshold value and high costs during routine clinical practice [9]. Nevertheless, inflammatory markers from routine complete blood count (CBC) are simple, readily available, and cost-effective and have been investigated in many diseases, including PCOS. Some of the studied markers include neutrophil/lymphocyte ratio (NLR), platelet/lymphocyte ratio (PLR), and white blood cell (WBC) count [10,11].
In recent years, delta neutrophil index (DNI), which reflects the ratio of circulating immature granulocytes (IG) to the total neutrophil count and can be detected by automatic hematology analyzers through the latest technological developments, has become a marker of infection and inflammation [12]. IG describe the myelocytes, promyelocytes, and metamyelocytes (neutrophil precursors) found in the bone marrow after the neonatal period. These immature neutrophils enter the circulation during infection, and DNI may have predictive and prognostic value in many different infectious conditions, such as acute appendicitis, bacterial peritonitis, and sepsis [13-16].
Additionally, DNI was identified as a prognostic marker for cardiac arrest and pulmonary embolism. In conclusion, DNI has prognostic importance in infectious diseases and diseases associated with systemic and sterile inflammation [17,18]. Here, we investigated the importance of DNI in the diagnosis of PCOS, in which inflammation is an important etiology. Discovering new simple and inexpensive inflammatory biomarkers for diagnosing PCOS in a clinical setting may contribute to disease follow-up. In addition, in PCOS patients, early detection and prevention of the inflammatory state may be beneficial for avoiding long-term risks such as diabetes and atherosclerosis [4]. Although the increased inflammatory response in patients with PCOS has been evaluated for various markers in the literature, the relationship between DNI, a new inflammatory marker, and PCOS has not been studied.
The presented retrospective clinical study was carried out between October 2019 and July 2022 at Afyonkarahisar Health Sciences University, Faculty of Medicine, Department of Obstetrics and Gynecology. The study was approved by Afyonkarahisar Health Sciences University Clinical Research Ethics Committee Medical Ethics Committee (2021/508).
Medical records of 227 patients were reviewed retrospectively, and clinical and laboratory data were obtained. According to the PCOS-rotterdam diagnostic criteria recommended by the European Society of Human Reproduction and Embryology, 72 patients diagnosed with PCOS who met at least two of the following criteria were included in the study: oligo and/or anovulation, clinical and/or biochemical findings of hyperandrogenism, and polycystic appearance [2]. Patients with hyperandrogenemia or other etiologies of ovulatory dysfunction (congenital adrenal hyperplasia, 21-hydroxylase deficiency, androgen-secreting tumors, Cushing’s syndrome, thyroid disease, and hyperprolactinemia) were excluded. Individuals with a known history of active infection in the last month or any autoimmune, inflammatory, metabolic, cardiovascular, hepatorenal, or malignant disease; menopausal patients; those with a deficiency in CBC parameters; and those who were pregnant were excluded from the study. Altogether, 155 non-pregnant white women, age-matched patients, aged 18-45 years with regular menstrual cycles and ovaries and a normal appearance on ultrasonography comprised the control group. The total blood count parameters obtained from peripheral venous blood sample taken in the morning on the third day of the menstrual cycle from all participants were scanned and recorded using the hospital’s patient data system. DNI parameters were analyzed using a Sysmex XE-2100 hematology analyzer (Sysmex Corporation, Kobe, Japan). This system is a flow cytometry-based hematological analyzer that uses two independent WBC counting methods: a myeloperoxidase (MPO) channel and a lobularity/nuclear density channel. DNI was determined using the following formula: DNI (leukocyte subfraction assayed in the MPO channel by cytochemical reaction)-(leukocyte subfraction assayed using the nuclear lobularity channel by reflected light beam measurements) [15].
The reference range for the DNI (IG: 103/μL) value measured in the automatic CBC measuring device (Sysmex XE-2100 hematology analyzer, Sysmex Corporation) used in the biochemistry laboratory of our university was 0-0.06×103/μL).
The primary outcome of this study was to examine the presence of a difference in the DNI between the PCOS and control groups, and the secondary outcome was to analyze the sensitivity and specificity of the DNI for PCOS.
Continuous variables are presented as means and standard deviations (SDs). Continuous variables between groups were compared using Student’s t-test or Mann-Whitney U-test, depending on the normality of the distribution. A receiver operating characteristic (ROC) curve analysis was performed to determine the appropriate cut-off points for individual indicators and to calculate sensitivity and specificity. The optimal significance cut-off value was calculated using Youden’s index.
This study included 72 PCOS patients and 155 healthy controls (n=227). No significant difference in mean age was observed between the two groups (mean±SD, patient: 27.46±7.66, control: 26.64±6.01; P=0.425). DNI and WBC count were significantly higher in the PCOS group than in the control (C) group, and red blood cell distribution width (RDW) was lower in the PCOS group than in the control group (DNI, patient: 0.0336±0.032, control: 0.0245±0.022; P=0.028; WBC, patient: 8.51±3.10, control: 7.46±2.08; P=0.011; RDW, patient: 23.05±13.73, control: 34.31±44.97; P=0.005). No significant differences in terms of other complete blood count parameters (neutrophils, lymphocytes, platelets, NLR, mean platelet volume [MPV]) and BMI (P>0.05) were observed between the two groups (Table 1). In order to understand the effect of obesity, obese (O) (BMI ≥25 kg/m2) and non-obese (NO) (BMI <25 kg/m2) and PCOS and C groups were compared. The number of neutrophils was significantly higher in the obese-PCOS (O-PCOS) group than in the obese-control (OC) group (P=0.016). Between the non-obese-PCOS (NO-PCOS) and non-obese-control (NO-C) groups, DNI and WBC were significantly higher in NO-PCOS patients, whereas RDW was significantly lower (P=0.018, 0.041, 0.001; respectively) (Table 2). The cut-off values were 0.015 for DNI, area under the curve (AUC)=0.588, significant (P=0.034; 95% confidence interval [CI], 0.509-0.667; sensitivity, 78%; specificity, 35%; Youden’s index=0.133); 9.35 for WBC, AUC=0.594, significant (P=0.022; 95% CI, 0.515-0.674; sensitivity, 32%; specificity, 86%; Youden’s index=0.171) for WBC; and 5.38 for neutrophils, AUC=0.628 was significant (P=0.002; 95% CI, 0.550-0.706; sensitivity, 39%; specificity, 81%; Youden’s index=0.202) (Table 3, Fig. 1). The ROC analysis for RDW was not significant (AUC=0.524; P=0.563). To our knowledge, no other studies on the association between PCOS and DNI have been reported in the literature.
In our study, the DNI was higher in the PCOS group than in the control group (P<0.05). In addition, when O-PCOS and NO-PCOS patients were considered separately, similar results were observed in the NO-PCOS group, whereas in the O-PCOS group, no significant difference with the controls was observed, suggesting that the inflammation detected in PCOS may be caused by the disorder itself rather than by obesity. Although the sensitivity and specificity were not at the desired levels for the DNI in the ROC analysis, the result was significant at a cut-off value of 0.015 (sensitivity, 78%; specificity, 35%; P=0.034). Our study is the first to show an increased DNI in patients with PCOS.
Although many mechanisms have been suggested for the development of PCOS, a single determinant and definitive treatment approach have not been revealed. However, recent studies have reported that the concentrations of many inflammatory markers are increased in PCOS patients. Some of these include intercellular adhesion molecule-1, TNF-α, and monocyte chemoattractant protein-1. Additionally, serum IL-6 levels are higher in women with PCOS than in controls [19]. Despite being studied in the bloodstream, the analysis of these molecules is difficult in routine clinical practice owing to precise threshold calculations and high costs [9]. However, the DNI, which was significant in our study, was obtained from a complete blood count. Neutrophils play a role in innate immunity and have more functions than antimicrobial responses in various tissues under pathological conditions [20].
Systemic inflammation leads to the destruction of circulating mature neutrophils and loss of active neutrophils. To compensate for this situation, the number of immature neutrophils (metamyelocytes, myelocytes, and promyelocytes) in the circulation increases, and a left shift occurs where the immature/total granulocyte ratio increases, which is an indicator of sepsis and inflammation [21]. Therefore, the DNI has been studied as a marker for many inflammatory and infectious diseases [15,16,22]. In addition to being reported as a diagnostic tool that better predicts mortality during sepsis than CRP [15], it was shown that it could predict perforation in patients with appendicitis [23]. The DNI has also been studied in obstetric patients. Serum DNI values were higher in women with severe preeclampsia than in women with normal pregnancies or mild preeclampsia [24]. In another study, DNI was identified as a predictive marker of histological chorioamnionitis in patients with preterm premature rupture of membranes [25]. In other studies, a higher DNI has been reported as a prognostic marker of conditions such as cardiac arrest and pulmonary embolism. Based on these studies, DNI values may reflect the severity of the infection and the severity of diseases associated with systemic and sterile inflammation in the absence of infection [17,18]. Since PCOS is known to be associated with an inflammatory response and DNI is increased during inflammation, DNI was significantly higher in both the PCOS and NO-PCOS patients in our study. No significant differences were observed according to OC in O-PCOS patients. Yilmaz et al. [26] have also reported significantly higher levels of NLR and MPV in PCOS patients; however, no significant difference was observed in O-PCOS patient. In another study, the PLR was significantly higher in NO-PCOS patients than in O-PCOS and NO-C patients [10]. Consistent with these studies, our results support obesity-independent inflammation in PCOS patients.
Yilmaz et al. [26] have reported higher levels of neutrophils, basophils, NLR, and MPV in lean patients with PCOS than in controls despite similar CRP levels, and these new markers may be more predictive of PCOS than CRP. By contrast, no significant difference in the NLR and MPV in PCOS patients in our study; however, we revealed DNI as a new marker for PCOS, although we could not compare it with CRP levels. In another study, an increased leukocyte count was accompanied by neutrophilia in PCOS, independent of BMI, consistent with our study, and this supported the presence of inflammation in PCOS [27]. Dasanu et al. [28] have also reported that thrombocytosis was not correlated with CRP in the complete blood count of PCOS patients. In our study, no significant difference was observed in the platelet counts of PCOS patients. Kabil Kucur et al. [29] have reported higher MPV levels in patients with PCOS and observed a significant decrease in MPV levels after treatment with ethyl estradiol/cyproterone acetate or metformin, suggesting that the underlying inflammation is important in PCOS and that anti-inflammatory treatment regimens can be beneficial. The DNI may show promise as a more sensitive parameter than these markers. However, further studies are required to understand the effects of treatment regimens on DNI.
Several studies have reported increased leukocyte counts in patients with PCOS [27,30-34]. However, the results on this subject are contradictory. A meta-analysis concluded that the increase in WBC and CRP levels in PCOS is independent of obesity [9]. Several studies have emphasized that increased leukocyte levels in PCOS were associated with increased neutrophil levels [27,35]. Several other studies have observed no difference in WBC counts between BMI-matched PCOS patients and controls [11,36,37]. In our study, significant neutrophilia and leukocytosis were observed in the O-PCOS and NO-PCOS groups, respectively.
Some organs such as the liver and adipose tissue contain few neutrophils under normal homeostatic conditions. However, neutrophil counts increased, and a metabolic imbalance occurred in the inflammatory environment caused by experimental obesity. Neutrophils secrete elastase from the azurophilic granules. This enzyme can destroy insulin receptor substrate 1 in adipocytes and hepatocytes, inducing insulin resistance and lipogenesis [38]. This neutrophil subtype and altered levels of elastase or its inhibitor (α1-antitrypsin) levels are associated with the onset of metabolic syndrome and diabetes [39]. In our study, the neutrophil levels were significantly higher in O-PCOS patients than those with O-C, supporting the function of neutrophils (P=0.016). In addition, stimulation of the proliferation and differentiation of hematopoietic cells, including T cells, as a result of increased leptin concentration and leptin resistance in women with obesity may contribute to neutrophilia [40,41]. In our study, patients with NO-PCOS also had increased leukocytosis. This may occur in NO-PCOS patients because of the contribution of hyperandrogenism to inflammation [42]. The difference in results between studies may be because central and simple obesity do not always coexist, and most studies simply used BMI rather than waist circumference measurements [43].
The hypothesis that increased inflammatory activity in PCOS is attributed to PCOS itself, independent of adiposity, awaits clarification [44]. As the amount of data supporting this situation increases, preventive treatment strategies may come to the fore.
The study’s results support the role of inflammatory status in PCOS, which remains controversial. The DNI can also be used as a cost-effective new indicator in patients who cannot be definitively diagnosed using the Rotterdam criteria. Further well-designed prospective studies are required to determine whether the DNI plays a role in evaluating the severity of inflammation in PCOS.
This study has some limitations. First, it was a retrospective study conducted at a single center. The DNI was calculated for each patient from a single blood sample; therefore, we did not determine the changes over time. In our clinic, automatic IG count parameters were obtained after 2018, and the number of patients was relatively limited as some patients’ BMI measurements were not recorded in their patient files. A comparison was not possible because inflammatory parameters such as CRP level were not routinely checked in this patient group. No waist or hip measurements indicated visceral obesity. Moreover, confirming that there were no chronic inflammatory conditions in the control group was difficult, with the leukocyte and neutrophil values in the complete blood count data within the normal reference range and the definition of the healthy control group.
The first investigation of DNI in patients with PCOS was a strength of this study.
PCOS is one of the most common endocrinopathies in women of reproductive age. Inflammation plays an important role in the etiology of PCOS. DNI values may reflect both the severity of the infection and the severity of diseases associated with systemic and sterile inflammation in the absence of infection. Therefore, DNI can also be used as a parameter that supports inflammation in PCOS and as a cost-effective marker.
Notes
Ethical approval
All procedures involving human participants were performed according to the ethical standards of the institution and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Fig. 1
ROC analyses of DNI, leukocytes, and neutrophils for prediction of PCOS. ROC, receiver operating characteristic; DNI, delta neutrophil index; PCOS, polycystic ovary syndrome.
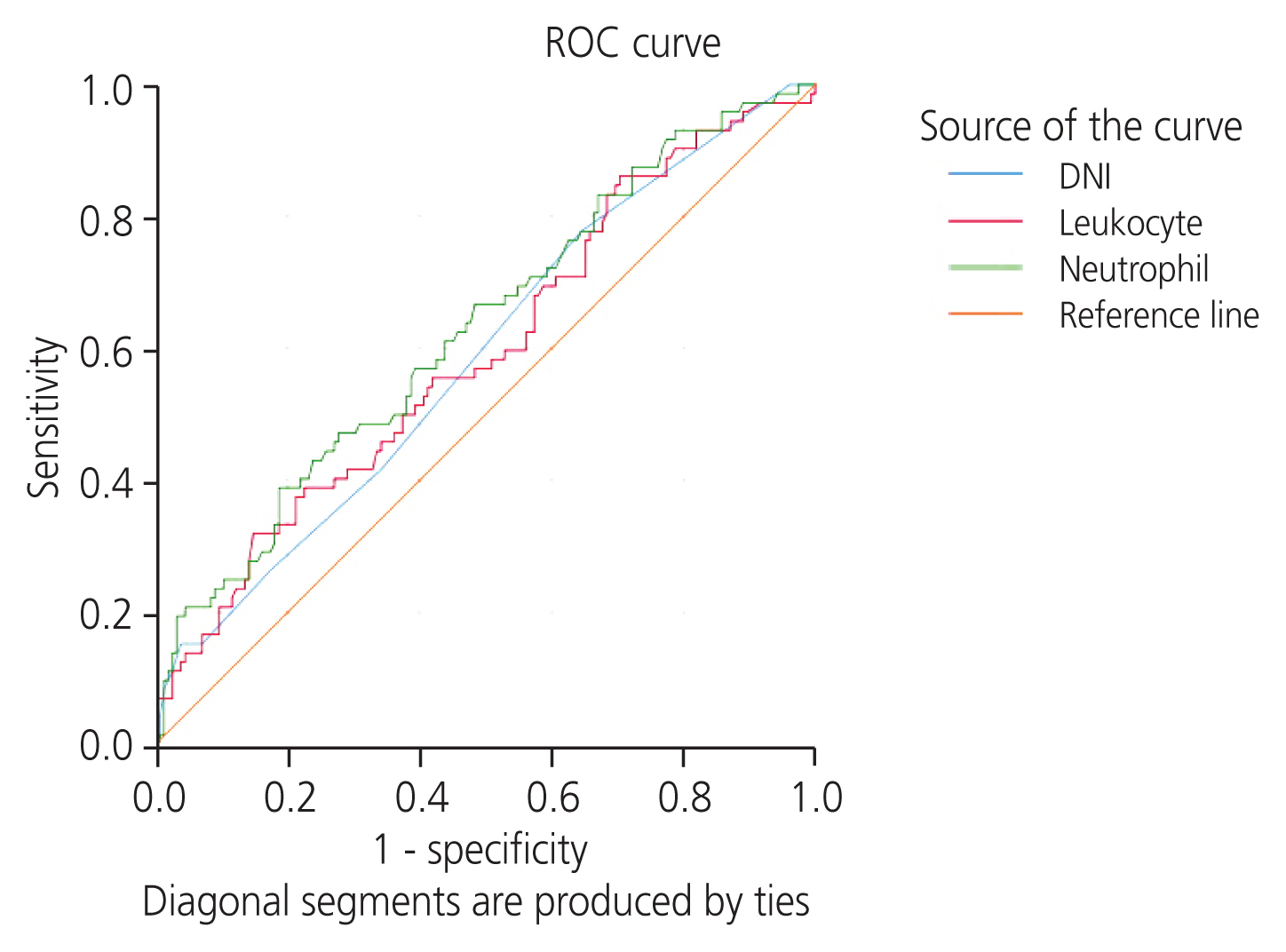
Table 1
Comparison of inflammatory markers between the PCOS and control groups
| PCOS patients (n=72) | Control group (n=155) | P | |
|---|---|---|---|
| DNI (IG: 103/μL) | 0.0336±0.032 | 0.0245±0.022 | 0.028a) |
| WBC (103/μL) | 8.51±3.10 | 7.46±2.08 | 0.011b) |
| Neutrophil (103/μL) | 5.42±2.73 | 4.46±3.39 | 0.037b) |
| RDW | 23.05±13.73 | 34.31±44.97 | 0.005b) |
| Lymphocyte (103/μL) | 2.33±0.69 | 2.18±0.67 | 0.127b) |
| NLR | 2.52±1.85 | 2.33±2.55 | 0.586b) |
| Platelets (103/μL) | 280.56±67.50 | 286.94±69.47 | 0.516b) |
| MPV (fL) | 10.37±0.88 | 10.43±0.93 | 0.659b) |
| Age (yr) | 27.46±7.66 | 26.64±6.01 | 0.425b) |
| BMI (kg/m2) | 25.29±2.79 | 25.47±3.10 | 0.947a) |
Table 2
Comparison of inflammatory markers between the PCOS and control groups
| O-PCOS (n=31) | O-control (n=68) | P-value | NO-PCOS (n=41) | NO-control (n=87) | P-value | |
|---|---|---|---|---|---|---|
| DNI (IG: 103/μL) | 0.029±0.024 | 0.027±0.029 | 0.719a) | 0.037±0.03 | 0.022±0.015 | 0.018a) |
| WBC (103/μL) | 8.43±3.45 | 7.34± 2.01 | 0.124b) | 8.57±2.84 | 7.52±2.14 | 0.041b) |
| Neutrophil (103/μL) | 5.37±3.18 | 4.12±1.84 | 0.016b) | 5.45±2.36 | 4.72±4.21 | 0.306b) |
| RDW | 26.91±15.84 | 28.13±39.7 | 0.870b) | 20.13±11.23 | 39.15±48.4 | 0.001b) |
| Lymphocyte (103/μL) | 2.31±0.84 | 2.08±0.63 | 0.132b) | 2.34±0.57 | 2.25±0.69 | 0.504b) |
| NLR | 2.70±2.60 | 2.43±2.80 | 0.650b) | 2.38±0.99 | 2.26±2.36 | 0.755b) |
| Platelets (103/μL) | 273.71±67.07 | 282.18±71.43 | 0.577b) | 285.73±68.2 | 290.7±68.09 | 0.703b) |
| MPV (fL) | 10.45±0.98 | 10.38±0.83 | 0.742b) | 10.46±1.0 | 10.31±0.80 | 0.402b) |
| Age (yr) | 29.10±8.23 | 26.5±6.65 | 0.123b) | 26.2±7.04 | 26.8±5.5 | 0.624b) |
| BMI (kg/m2) | 27.53±2.77 | 28.4±2.2 | 0.120a) | 23.61±1.16 | 23.23±1.36 | 0.121a) |
Table 3
Comparison of the ROC analyses of markers (DNI, WBC, and neutrophil) for prediction of PCOS
| Marker | AUC | Sensitivity (%) | Specificity (%) | Cut-off | 95% CI | Youden’s index | P-valuea) | |
|---|---|---|---|---|---|---|---|---|
| Lower bound | Upper bound | |||||||
| DNI | 0.588 | 78.0 | 35.0 | 0.015 | 0.509 | 0.667 | 0.133 | 0.034 |
| WBC | 0.594 | 32.0 | 86.0 | 9.35 | 0.515 | 0.674 | 0.171 | 0.022 |
| Neutrophil | 0.628 | 39.0 | 81.0 | 5.38 | 0.550 | 0.706 | 0.202 | 0.002 |
| RDW | 0.524 | 0.563 | ||||||
References
1. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab 2004;89:2745-9.


2. Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004;19:41-7.


3. Legro RS, Arslanian SA, Ehrmann DA, Hoeger KM, Murad MH, Pasquali R, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2013;98:4565-92.



4. Dandona P, Aljada A, Chaudhuri A, Bandyopadhyay A. The potential influence of inflammation and insulin resistance on the pathogenesis and treatment of atherosclerosis-related complications in type 2 diabetes. J Clin Endocrinol Metab 2003;88:2422-9.


5. Abraham Gnanadass S, Divakar Prabhu Y, Valsala Gopalakrishnan A. Association of metabolic and inflammatory markers with polycystic ovarian syndrome (PCOS): an update. Arch Gynecol Obstet 2021;303:631-43.


6. Guzelmeric K, Alkan N, Pirimoglu M, Unal O, Turan C. Chronic inflammation and elevated homocysteine levels are associated with increased body mass index in women with polycystic ovary syndrome. Gynecol Endocrinol 2007;23:505-10.


7. Wang J, Gong P, Li C, Pan M, Ding Z, Ge X, et al. Correlation between leptin and IFN-γ involved in granulosa cell apoptosis in PCOS. Gynecol Endocrinol 2020;36:1051-6.


8. Hatziagelaki E, Pergialiotis V, Kannenberg JM, Trakakis E, Tsiavou A, Markgraf DF, et al. Association between biomarkers of low-grade inflammation and sex hormones in women with polycystic ovary syndrome. Exp Clin Endocrinol Diabetes 2020;128:723-30.


9. Escobar-Morreale HF, Luque-Ramírez M, González F. Circulating inflammatory markers in polycystic ovary syndrome: a systematic review and metaanalysis. Fertil Steril 2011;95:1048-58e1. -2.



10. Çakıroğlu Y, Vural F, Vural B. The inflammatory markers in polycystic ovary syndrome: association with obesity and IVF outcomes. J Endocrinol Invest 2016;39:899-907.


11. Shi Y, Han T, Cui L, Wu G, Zheng R, Xia M, et al. White blood cell differential counts in patients with polycystic ovary syndrome: a pilot study on Chinese women. Eur J Obstet Gynecol Reprod Biol 2013;170:162-4.


12. Kratz A, Maloum K, O’Malley C, Zini G, Rocco V, Zelmanovic D, et al. Enumeration of nucleated red blood cells with the ADVIA 2120 hematology system: an international multicenter clinical trial. Lab Hematol 2006;12:63-70.


13. Nierhaus A, Klatte S, Linssen J, Eismann NM, Wichmann D, Hedke J, et al. Revisiting the white blood cell count: immature granulocytes count as a diagnostic marker to discriminate between SIRS and sepsis--a prospective, observational study. BMC Immunol 2013;14:8.



14. Kim OH, Cha YS, Hwang SO, Jang JY, Choi EH, Kim HI, et al. The use of delta neutrophil index and myeloperoxidase index for predicting acute complicated appendicitis in children. PLoS One 2016;11:e0148799.



15. Seok Y, Choi JR, Kim J, Kim YK, Lee J, Song J, et al. Delta neutrophil index: a promising diagnostic and prognostic marker for sepsis. Shock 2012;37:242-6.

16. Lim TS, Kim BK, Lee JW, Lee YK, Chang S, Kim SU, et al. Use of the delta neutrophil index as a prognostic factor of mortality in patients with spontaneous bacterial peritonitis: implications of a simple and useful marker. PLoS One 2014;9:e86884.



17. Kong T, Park YS, Lee HS, Kim S, Lee JW, Yu G, et al. Value of the delta neutrophil index for predicting 28-day mortality in patients with acute pulmonary embolism in the emergency department. Shock 2018;49:649-57.


18. Yune HY, Chung SP, Park YS, Chung HS, Lee HS, Lee JW, et al. Delta neutrophil index as a promising prognostic marker in out of hospital cardiac arrest. PLoS One 2015;10:e0120677.



19. Peng Z, Sun Y, Lv X, Zhang H, Liu C, Dai S. Interleukin-6 levels in women with polycystic ovary syndrome: a systematic review and meta-analysis. PLoS One 2016;11:e0148531.



20. Rosales C. Neutrophil: a cell with many roles in inflammation or several cell types? Front Physiol 2018;9:113.



21. Ansari-Lari MA, Kickler TS, Borowitz MJ. Immature granulocyte measurement using the sysmex XE-2100. Relationship to infection and sepsis. Am J Clin Pathol 2003;120:795-9.


22. Kim H, Kim Y, Lee HK, Kim KH, Yeo CD. Comparison of the delta neutrophil index with procalcitonin and C-reactive protein in sepsis. Clin Lab 2014;60:2015-21.


23. Shin DH, Cho YS, Kim YS, Ahn HC, Oh YT, Park SO, et al. Delta neutrophil index: a reliable marker to differentiate perforated appendicitis from non-perforated appendicitis in the elderly. J Clin Lab Anal 2018;32:e22177.



24. Cho HY, Jung I, Kim SJ, Park YW, Kim YH, Kwon JY. Increased delta neutrophil index in women with severe preeclampsia. Am J Reprod Immunol 2017;78:e12705.

25. Cho HY, Jung I, Kwon JY, Kim SJ, Park YW, Kim YH. The delta neutrophil index as a predictive marker of histological chorioamnionitis in patients with preterm premature rupture of membranes: a retrospective study. PLoS One 2017;12:e0173382.



26. Yilmaz MA, Duran C, Basaran M. The mean platelet volume and neutrophil to lymphocyte ratio in obese and lean patients with polycystic ovary syndrome. J Endocrinol Invest 2016;39:45-53.


27. Herlihy AC, Kelly RE, Hogan JL, O’Connor N, Farah N, Turner MJ. Polycystic ovary syndrome and the peripheral blood white cell count. J Obstet Gynaecol 2011;31:242-4.


28. Dasanu CA, Clark BA 3rd, Ichim TE, Alexandrescu DT. Polycystic ovary syndrome: focus on platelets and prothrombotic risk. South Med J 2011;104:174-8.


29. Kabil Kucur S, Gozukara I, Aksoy A, Uludag EU, Keskin H, Kamalak Z, et al. How medical treatment affects mean platelet volume as a cardiovascular risk marker in polycystic ovary syndrome? Blood Coagul Fibrinolysis 2015;26:862-5.


30. Margolis KL, Manson JE, Greenland P, Rodabough RJ, Bray PF, Safford M, et al. Leukocyte count as a predictor of cardiovascular events and mortality in postmenopausal women: the women’s health initiative observational study. Arch Intern Med 2005;165:500-8.


31. Keskin Kurt R, Okyay AG, Hakverdi AU, Gungoren A, Dolapcioglu KS, Karateke A, et al. The effect of obesity on inflammatory markers in patients with PCOS: a BMI-matched case-control study. Arch Gynecol Obstet 2014;290:315-9.


32. Ruan X, Dai Y. Study on chronic low-grade inflammation and influential factors of polycystic ovary syndrome. Med Princ Pract 2009;18:118-22.


33. Phelan N, O’Connor A, Kyaw Tun T, Correia N, Boran G, Roche HM, et al. Leucocytosis in women with polycystic ovary syndrome (PCOS) is incompletely explained by obesity and insulin resistance. Clin Endocrinol (Oxf) 2013;78:107-13.


34. Papalou O, Livadas S, Karachalios A, Tolia N, Kokkoris P, Tripolitakis K, et al. White blood cells levels and PCOS: direct and indirect relationship with obesity and insulin resistance, but not with hyperandogenemia. Hormones (Athens) 2015;14:91-100.


35. Ibáñez L, Jaramillo AM, Ferrer A, de Zegher F. High neutrophil count in girls and women with hyperinsulinaemic hyperandrogenism: normalization with metformin and flutamide overcomes the aggravation by oral contraception. Hum Reprod 2005;20:2457-62.


36. Silfeler DB, Kurt RK, Yengil E, Un B, Arica S, Baloglu A. Evaluation of mean platelet volume values in lean women with polycystic ovary syndrome. Pak J Med Sci 2014;30:589-92.


37. Tola EN, Yalcin SE, Dugan N. The predictive effect of inflammatory markers and lipid accumulation product index on clinical symptoms associated with polycystic ovary syndrome in nonobese adolescents and younger aged women. Eur J Obstet Gynecol Reprod Biol 2017;214:168-72.


38. Talukdar S, Oh DY, Bandyopadhyay G, Li D, Xu J, McNelis J, et al. Neutrophils mediate insulin resistance in mice fed a high-fat diet through secreted elastase. Nat Med 2012;18:1407-12.



39. Mansuy-Aubert V, Zhou QL, Xie X, Gong Z, Huang JY, Khan AR, et al. Imbalance between neutrophil elastase and its inhibitor α1-antitrypsin in obesity alters insulin sensitivity, inflammation, and energy expenditure. Cell Metab 2013;17:534-48.



40. Lord GM, Matarese G, Howard JK, Baker RJ, Bloom SR, Lechler RI. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 1998;394:897-901.


41. van Rossum EF, Nicklas BJ, Dennis KE, Berman DM, Goldberg AP. Leptin responses to weight loss in postmenopausal women: relationship to sex-hormone binding globulin and visceral obesity. Obes Res 2000;8:29-35.


42. Gilliver SC, Ashworth JJ, Mills SJ, Hardman MJ, Ashcroft GS. Androgens modulate the inflammatory response during acute wound healing. J Cell Sci 2006;119:722-32.


- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 1,432 View
- 107 Download
- Related articles in Obstet Gynecol Sci
-
A case of endometrial cancer in a young patient with polycystic ovary syndrome.2002 September;45(9)
Recent Advances in the Management of Polycystic Ovary Syndrome.2005 August;48(8)




















