|
 |
- Search
| Obstet Gynecol Sci > Volume 60(3); 2017 > Article |
Abstract
A 30-year-old woman experienced severe abdominal pain 8 days after vaginal delivery. The patient was diagnosed with hemoperitoneum due to rupture of the left uterine artery pseudoaneurysm, which was confirmed via ultrasound with color Doppler and computed tomography scans. This patient was treated with bilateral uterine artery embolization to maintain fertility. A uterine artery pseudoaneurysm that causes delayed postpartum hemorrhage can occur after cesarean section or vaginal delivery. A uterine artery pseudoaneurysm can be fatal, so its detection and diagnosis are critical. Herein, we report a case of delayed postpartum hemoperitoneum due to uterine artery pseudoaneurysm rupture.
Delayed postpartum hemorrhage occurs between 24 hours and 12 weeks postpartum. Delayed postpartum hemorrhage may be caused by various factors, such as retained products of conception, infection, abnormal involution of the placental site or uterine artery pseudoaneurysm (UAP) [1]. When a punctured or lacerated artery is not completely sealed, blood may escape and diffuse into adjacent tissues and collect in perivascular areas. If the blood collection maintains a link with the parent vessel, it may cause a pseudoaneurysm. A pseudoaneurysm boundary forms with the peripheral thrombus and part of the three arterial layers [2]. UAP is rare, although a recent study reported that it occurs at an actual ratio of 2 to 3 instances per 1,000 deliveries [3]. UAP may occur as a consequence of intra-cesarean section vascular trauma, traumatic abortion/delivery or uncomplicated spontaneous vaginal delivery [3]. UAP often leads to a delayed postpartum hemorrhage that ruptures within the uterine cavity. However, there was few report of case in which muscle layer rupture was the cause of postpartum hemoperitoneum. Therefore, we report a case of delayed postpartum hemoperitoneum caused by intramyometrial pseudoaneurysm rupture after vaginal delivery.
A 30-year-old woman (gravida 2, para 1) visited the emergency department on postpartum day 8 with lower abdominal cramping as her major complaint. She had a normal spontaneous vaginal delivery at 39 weeks of pregnancy. When she arrived at the emergency department, her status was stable. Her blood pressure was 120/70 mmHg; pulse rate was 78 beats/min; and respiration rate was 20 breaths/min. Blood tests revealed normal outcomes, including hemoglobin (10.3 g/dL) and platelet (308,000/┬ĄL) measurements. She had no remarkable medical or surgical history, except one spontaneous abortion without a coagulation disorder.
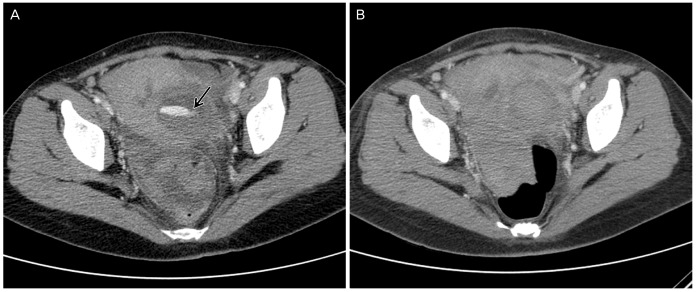
Via an ultrasound examination, a 53├Ś44├Ś44-mm-size pulsating hypoechoic cyst was identified in the myometrium of the left lower segment (Fig. 1A). Based on a color Doppler examination, turbulent artery-like flow with a ŌĆśto-and-froŌĆÖ pattern was observed (Fig. 1B). In the pelvis, a small hematoma (mildly echogenic fluid) without para gutter fluid collection was present. Computed tomography (CT) scanning was performed, and the patient was diagnosed with hemoperitoneum. The CT scan (intra-abdominal and pelvic cavity) revealed high density fluid collection and leakage of contrast media into the left side of the uterus (Fig. 2A). While undergoing the CT scan, she complained of pain aggravation, and her blood pressure decreased to 100/60 mmHg. Her pulse rate had increased to 98 beats/min. In a blood test conducted 2 hours later, her hemoglobin level had dropped to 6.5 g/dL. Bleeding of the left uterine artery was confirmed via arteriography, which was conducted based on the suspicion of pseudoaneurysm. Non-invasive, transcatheter arterial embolization (TAE) was planned as a treatment to preserve the patient's fertility. After confirming the leakage of contrast media and hypervascularity at the distal branch of the left uterine artery, TAE was performed using gelfoam. Her right uterine artery was also hypertrophied and was also treated with TAE using gelfoam. We confirmed that there was no contrast media leakage suggestive of active bleeding from the follow-up CT scan one day later (Fig. 2B). She was discharged 3 days later. However, she revisited the hospital 2 weeks later with lower abdominal pain as her major complaint. We conducted a physical exam and CT scan. She was diagnosed with uterine hematoma infection, and antibiotics were administered. Based on the ultrasonic examination that was conducted 3 months later, the intramyometrial hematoma was reduced to 3 cm.
Delayed postpartum hemorrhage is defined as postdelivery hemorrhage that can occur from 24 hours to 12 weeks postpartum [1]. UAP is a rare cause of delayed postpartum hemorrhage and can occur after dilatation and curettage, cesarean section or uncomplicated spontaneous vaginal delivery [3]. Unlike a true aneurysm, a pseudoaneurysm is not wrapped by three layers of the arterial wall. Pseudoaneurysm is a complication due to local trauma that is accompanied by vascular injury. Blood flow out of the damaged vessels collects in the perivascular area and is surrounded by adjacent tissues. The blood then forms a pseudoaneurysm that is in communication with the parent vessel [2]. As reported previously, postpartum hemorrhage can occur as a consequence of pseudoaneurysm leakage-induced vaginal bleeding. However, in the present case, hemoperitoneum had occurred as a consequence of pseudoaneurysm rupture into the intra-abdominal space. The cause of this rupture was postpartum pseudoaneurysm and hemoperitoneum that had formed 8 days after delivery. Thus, this case should be categorized as delayed postpartum hemorrhage.
The clinical prognoses of UAP are diverse. For example, this condition can be asymptomatic, cause pain, exert a mass effect on the adjacent neurovascular bundle, bleed, rupture, thrombose, or cause distal embolization [4]. In a review, 32 cases were analyzed, and the size of the pseudoaneurysm was measured. The mean size of the pseudoaneurysms was 13┬▒7 mm (mean┬▒standard deviation), and most pseudoaneurysms were Ōēż20 mm (84%) [2,5,6,7,8,9,10,11,12,13]. Among the 32 described cases, 2 were diagnosed as intra-myometrial UAPs. These cases were characterized by severe abdominal pain and relatively large pseudoaneurysm sizes (30 and 35 mm) [2,13]. In our patient, sudden severe abdominal pain occurred without vaginal bleeding. The pseudoaneurysm size (measured via ultrasonic examination) was rather large (53├Ś44├Ś44 mm). Unlike other cases, the lateral uterine muscle was ruptured and had progressed to hemoperitoneum. During a pseudoaneurysm, the tissues surrounding the ruptured parent artery act as a wall for the aneurysm. In most cases, the pseudoaneurysm is most likely to form on the internal weak side of the uterus, and part of the 3-layer artery and thrombus would act as the aneurysm wall. Subsequently, the pseudoaneurysm might rupture at a relatively small size (allowing the blood to flow out and resulting in vaginal bleeding). In cases of intramyometrial UAP, the myometrium is solid. Thus, the pseudoaneurysm persists without rupture until it grows to a relatively large size and causes severe pain due to increased intramural pressure.
Pseudoaneurysm can be diagnosed using color Doppler, ultrasonography, CT and angiography. For UAP, there is no established optimized method for its diagnosis because it is a rare condition. In cases of postpartum pseudoaneurysm, ultrasonography is the first method utilized during either a routine postpartum exam or treatment for postpartum hemorrhage. Uterine pseudoaneurysm can be confirmed by observing a pulsating hypoechoic mass connected to a parent artery with a narrow neck. Color Doppler scans can reveal the ŌĆśto-and-froŌĆÖ pattern and allows differentiation from other vascular abnormalities, such as arteriovenous malformation or fistula [11]. A CT scan or angiography can often help to determine the pathological origin [8].
In the present case, a small hematoma was observed (via ultrasonography) inside her pelvis. On the CT scan conducted 1 hour later, the hemoperitoneum had worsened, and we observed fluid collection in the deep subphrenic space. After the uterine muscles ruptured, massive bleeding occurred for a short time, which required emergency treatment. The patient's blood pressure decreased and pulse rate increased. Nevertheless, a non-surgical method (uterine artery embolization) was performed. The patient wanted to maintain her fertility, and TAE was performed successfully. This result indicates that TAE is a safe and effective technique for the treatment of UAP rupture-induced severe postpartum bleeding [6,12]. There are no randomized controlled trials to guide management of postpartum hemorrhage. But TAE is now well-accepted procedure for the management of postpartum hemorrhage [14]. Indications for TAE in women with postpartum hemorrhage include uterine atony, UAP, invasive placentation and genital tears [14]. TAE should be preferred to other techniques after vaginal birth and in secondary postpartum hemorrhage [14]. Surgery is preferred when failure of conservative treatments and complex uterine rupture are suspected [14]. The first-line treatment for UAP is TAE because it is highly effective and avoids the potential complications of emergent hysterectomy [8]. When conducting TAE, recurrent bleeding is likely to occur if unilateral embolization is implemented. Thus, it is important to perform bilateral embolization [15]. Bilateral embolization is more effective than unilateral embolization and it does not increase the risk for future infertility [5]. This procedure confirms the effects of embolization for intramyometrial UAP rupture-induced hemoperitoneum.
We conclude that a large intramyometrial pseudoaneurysm is accompanied by pain. The potential for rupture is present during the postpartum period and can cause vaginal bleeding and hemoperitoneum. Detection and diagnosis of intramyometrial UAP, which can be fatal, is essential. TAE should be implemented immediately. Bilateral embolization is a safe and effective treatment method for UAP rupture-induced hemoperitoneum.
References
1. Cunningham FG, Leveno KJ, Bloom SL, Spong CY, Dashe JS, Hoffman BL, et al. Williams obstetrics. 24th ed. New York (NY): McGrawHill; 2014.
2. Zimon AE, Hwang JK, Principe DL, Bahado-Singh RO. Pseudoaneurysm of the uterine artery. Obstet Gynecol 1999;94:827-830. PMID: 10546745.


3. Baba Y, Matsubara S, Kuwata T, Ohkuchi A, Usui R, Saruyama M, et al. Uterine artery pseudoaneurysm: not a rare condition occurring after non-traumatic delivery or non-traumatic abortion. Arch Gynecol Obstet 2014;290:435-440. PMID: 24691826.


4. Lee WK, Roche CJ, Duddalwar VA, Buckley AR, Morris DC. Pseudoaneurysm of the uterine artery after abdominal hysterectomy: radiologic diagnosis and management. Am J Obstet Gynecol 2001;185:1269-1272. PMID: 11717672.


5. Nanjundan P, Rohilla M, Raveendran A, Jain V, Khandelwal N. Pseudoaneurysm of uterine artery: a rare cause of secondary postpartum hemorrhage, managed with uterine artery embolisation. J Clin Imaging Sci 2011;1:14PMID: 21977387.


6. Soyer P, Fargeaudou Y, Morel O, Boudiaf M, Le Dref O, Rymer R. Severe postpartum haemorrhage from ruptured pseudoaneurysm: successful treatment with transcatheter arterial embolization. Eur Radiol 2008;18:1181-1187. PMID: 18270711.


7. Gondo S, Urushiyama D, Yoshizato T, Kora S, Maehara M, Kondo H, et al. The successful detection of postpartum unruptured vaginal pseudoaneurysm using ultrasonography: a case report. Springerplus 2014;3:482PMID: 25221737.



8. Kwon HS, Cho YK, Sohn IS, Hwang HS, Seo KJ, Park WI, et al. Rupture of a pseudoaneurysm as a rare cause of severe postpartum hemorrhage: analysis of 11 cases and a review of the literature. Eur J Obstet Gynecol Reprod Biol 2013;170:56-61. PMID: 23746797.


9. Yi SW, Lee JH. Uterine pseudoaneurysm leakage may cause delayed postpartum haemorrhage: multidetector CT with angiography and transcatheter uterine arterial embolisation. J Obstet Gynaecol 2012;32:552-555. PMID: 22779960.


10. Yun SY, Lee DH, Cho KH, Lee HM, Choi YH. Delayed postpartum hemorrhage resulting from uterine artery pseudoaneurysm rupture. J Emerg Med 2012;42:e11-e14. PMID: 21497477.


11. Gurses C, Yilmaz S, Biyikli S, Yildiz IO, Sindel T. Uterine artery pseudoaneurysm: unusual cause of delayed postpartum hemorrhage. J Clin Ultrasound 2008;36:189-191. PMID: 17722017.


12. McGonegle SJ, Dziedzic TS, Thomas J, Hertzberg BS. Pseudoaneurysm of the uterine artery after an uncomplicated spontaneous vaginal delivery. J Ultrasound Med 2006;25:1593-1597. PMID: 17121956.


13. Henrich W, Fuchs I, Luttkus A, Hauptmann S, Dudenhausen JW. Pseudoaneurysm of the uterine artery after cesarean delivery: sonographic diagnosis and treatment. J Ultrasound Med 2002;21:1431-1434. PMID: 12494988.


14. Soyer P, Dohan A, Dautry R, Guerrache Y, Ricbourg A, Gayat E, et al. Transcatheter arterial embolization for postpartum hemorrhage: indications, technique, results, and complications. Cardiovasc Intervent Radiol 2015;38:1068-1081. PMID: 25677130.


15. Cooper BC, Hocking-Brown M, Sorosky JI, Hansen WF. Pseudoaneurysm of the uterine artery requiring bilateral uterine artery embolization. J Perinatol 2004;24:560-562. PMID: 15329736.


-
METRICS

- Related articles in Obstet Gynecol Sci
-
A case of spontaneous hemoperitoneum by uterine vessel rupture in pregnancy2016 November;59(6)