 |
 |
- Search
| Obstet Gynecol Sci > Volume 58(5); 2015 > Article |
Abstract
Objective
Revascularization is critical for successful ovarian tissue transplantation. Vascular endothelial growth factor (VEGF) and angiopoietin-2 (angpt-2) are the principal mediators of neovascularization. This study was designed to assess VEGF and angpt-2 levels in cryopreserved ovarian tissue after heterotopic autotransplantation.
Methods
Ovarian tissues harvested from ICR mice at 5 to 6 weeks of age were stratified as follows: no cryopreservation (controls, group I); vitrification in VFS-40 (vitrification, group II); and gradual freezing in dimethyl sulfoxide (slow-freezing, group III). Frozen specimens were thawed at room temperature, assaying VEGF and angpt-2 levels 1 week after cryopreservation and 2 weeks after autotransplantation.
Results
VEGF and angpt-2 protein levels were significantly lower in cryopreserved ovaries of groups II and III than in controls (group I, P<0.05), whereas groups II and III did not differ significantly in this regard. After autotransplantation of cryopreserved ovarian tissue, VEGF and angpt-2 protein levels did not differ significantly by technique but tended to be lower than corresponding levels in controls.
Conclusion
Expression of angiogenic factors in ovarian tissue is thought to vary by method of cryopreservation. Our findings indicate that levels of angiogenic factors expressed in cryopreserved ovarian tissue after autotransplantation do not differ appreciably from control levels, regardless of cryopreservation technique.
The life expectancies of premenopausal women with cancer have increased greatly due to diagnostic and therapeutic advances. Subsequently, the population of long-term adolescent and adult survivors of childhood malignancies has grown [1]. Ovarian tissue banking is a promising option for preserving fecundity in young female cancer patients facing sterilization by chemotherapy and/or radiotherapy [2]. However, the limited functional duration of some nonvascularized ovarian grafts may be due in part to ischemic injury sustained until revascularization is adequate [3]. Revascularization may thus be a critical step in successful ovarian tissue transplantation.
Anatomic locations utilized in transplanting cryopreserved ovarian tissue have varied. Heterotopic sites such as the subcutaneous of the abdominal wall, or forearm and rectus muscle generally have functioned well as reservoirs for ovarian restoration [4,5,6]. For the present study, we chose the abdominal wall, given the temperature and blood flow benefits of a subcutaneous environment and the relative ease of surgical access [7]. The abdominal wall is also the logical choice in experimental mice.
A number of angiogenic factors have been identified as principal mediators of neovascularization. Vascular endothelial growth factor (VEGF) is an endothelial cell mitogen that regulates angiogenesis and blood vessel permeability in transplanted ovarian tissue [8,9,10,11]. In addition, angiopoietins are required to regulate vascular ingrowth and stability [11,12]. Angiopoietin-2 (angpt-2) enables endothelial cell migration and proliferation, further promoting angiogenesis in the presence of VEGF. In the absence of VEGF, angpt-2 blocks recruitment of periendothelial support cells, resulting in vascular destabilization and regression [13,14,15]. This study was aimed at investigating the effects of cryopreservation techniques (vitrification and slow-freezing) on ovarian expression of angiogenic factors by determining the levels of VEGF and angpt-2 in successful heterotopic ovarian autotransplants of mice.
Female ICR mice were purchased from Koatech (Pyeongtaek, Korea). The mice were housed under light-controlled and temperature-controlled conditions (12 hours of light and 12 hours of darkness, 22±2℃), and were provided with sterile food and water. Mice were treated in accordance with the standard guidelines for laboratory animal care at animal facility of the Gyeongsang National University (GLA-090107-M0001).
ICR mice (n=30) 5 to 6 weeks of age in three groups (10 each) were anesthetized, and bilateral oophorectomies were performed. Operative wounds were sutured using PolySorb 3-0 layer by layer. Harvested ovaries of the two test groups were subjected to cryopreservation, reserving one ovary of each pair for later autotransplantation (Fig. 1).
The specimens were stratified as follows: no cryopreservation (controls, group I); vitrification in VFS-40 (vitrification, group II); and gradual freezing in dimethyl sulfoxide (slow-freezing, group III).
Experimentation was conducted in two phases. First, angiogenic factors were evaluated in test ovaries (cryopreserved for one week) and in controls. Second, angiogenic factors were evaluated two weeks after autotransplantation of test ovaries. In the control group, non-cryopreserved ovaries were autotransplanted immediately following oophorectomies.
Paired ovaries were immersed 10 minutes in an equilibration solution (EG-20), consisting of 20% ethylene glycol in Dulbecco's phosphate-buffered saline (m-DPBS) with 10% fetal bovine serum (FBS). This was followed by a 2-minutes immersion in vitrification solution (EFS-40), consisting of 40% ethylene glycol (v/v), 18% Ficoll (w/v), 0.5 mol/L sucrose, and 20% FBS in m-DPBS. The specimens were then loaded onto an electron microscope grid and immediately quick-frozen in liquid nitrogen. After 1 week, a three-step cryoprotectant dilution method was implemented for thawing [16]. One ovary was used for Western blot analysis, the other ovary was reserved for autotransplantation.
Paired ovaries were transferred to cryovials containing a cryoprotective mixture (Leibovitz L-15 medium, 10% FBS, and 1.5 M dimethyl sulfoxide) for controlled freezing via planar cryochamber. Cooling began at 4℃, proceeding at 2℃/min (past induction of ice nucleation at -7℃) until reaching -35℃. Freezing was accelerated to 25℃/min, plunging the cryovials into liquid nitrogen once a threshold temperature (-140℃) was reached. After 1 week, thawing was carried out at room temperature.
One ovary of each harvested pair served as heterotopic autotransplant, to be done one week after cryopreservation. Following inhalation anesthesia, the animal's abdominal wall was carefully opened, creating space for a graft without breach of peritoneum. Ovaries were affixed to exposed peritoneum using PolySorb 3-0 suture. Two weeks later, the mice were sacrificed and the ovaries were recovered (Fig. 2). Follicles were observed under microscopy, and Western blot analysis was performed.
Harvested ovaries were preserved in Pro-prep (iNtRON Biotechnology, Seoul, Korea) solution until analysis. Proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis in 8% gels and blotted onto polyvinylidene fluoride membranes. The blots were blocked (1 hour) using 5% skimmilk in TBST (20 mM Tris-buffered saline and 0.05% Tween 20, pH 7.5) at room temperature for overnight incubation (4℃) with anti-VEGF antibody (1:500). Blots were washed for secondary antibody incubation, followed by washing and detection of immunoreactivity with enhanced chemiluminescence (Amersham ECL Reagent, GE Healthcare Biosciences, Pittsburgh, PA, USA). Data were independently generated in triplicate.
All data were expressed as mean±standard error. Statistical computations relied on standard software (PASW ver. 18.0, SPSS Inc., Chicago, IL, USA). Non-parametric Kruskal-Wallis method was applied for comparisons, using Tukey's test for post-hoc analysis. Significance was set at P<0.05.
Two weeks after autotransplantation, any viable ovarian tissue found under abdominal wall with observable follicles by microscopy was defined as a successful transplant (Fig. 2). The number of successfully transplanted ovaries was 8/10 in controls (group I), 6/10 with vitrification (group II) and 7/10 with slow-freezing (group III).
With cryopreservation, lower levels of VEGF and angpt-2 were expressed relative to controls (P<0.05), and levels did not differ significantly by cryopreservation method (Fig. 3).
Two weeks after autotransplantation, whole ovaries removed from mice were analyzed. VEGF and angpt-2 expression in cryopreservation groups trended lower than in controls, but the differences were not significant; and levels did not differ significantly by cryopreservation method (Fig. 4).
Banking of ovarian tissue is a promising technique for preservation of fecundity in young female cancer patients [17,18]. Although this strategy is still considered experimental, proof-of-principle has existed for some time. Earlier studies indicate that variable but large fractions of follicles are lost by ischemia while grafts revascularize during their first 2 to 5 days, whereas the impact of cryoinjury is comparatively minor [19,20]. Angiogenesis in frozen-thawed ovarian tissue of rats begins within 48 hours of transplantation, commensurate with increased angpt-2 and VEGF189 mRNA levels, but requires more than 7 days to reach completion [21]. By facilitating angiogenesis and thus limiting the duration of ischemia, reductions in necrotic tissue and fractions of wasted follicles should result, thereby prolonging graft function and potentiating fertility [22].
VEGF was initially credited with the ability to induce vascular leakage/permeability and to promote ingrowth of vessels [23]. Indeed, incubation of human ovarian tissue with VEGF has been shown to improve graft survival [24]. In addition, angpt-2 is required for regulating angiogenesis and for stability of blood vessels [12], as well as securing folliculogenesis during the preovulatory period [15]. Hence, VEGF and angpt-2 levels are important indices of successful cryopreserved ovarian transplants.
Our previous data and present findings have shown that VEGF and angpt-2 are diminished by cryopreservation, regardless of the method utilized. Although vitrification with EFS-40 proved more efficient than slow freezing for preserving angiogenic factors [16] in our earlier work, no differences between vitrification with EFS-40 and slow freezing were evident in the present investigation. Currently, we are unable to explain this discrepancy, other than to implicate a delay in conducting the Western blot analysis after autotransplantation of cryopreserved ovaries in this study.
Slow freezing of ovarian tissue preserves cell morphology and hormone production in the course of freeze-thaw maneuvers [25]. Vitrification also preserves follicular morphology/viability and stromal density [26]. On the other hand, observed disparities in angiogenic factors with cryopreservation (vs. controls) were largely reversed after autotransplantation. Revascularization of the grafts rapidly up-regulated gene expression of angiogenic factors [27]. In this instance, observed increases in angiogenic factors of test specimens approached levels found in non-cryopreserved controls.
Although the cryopreserved ovarian autotransplants herein are considered successful, tissue necrosis was ample, and reduction in normal tissue was substantial. Apparently, VEGF and angpt-2 disruption by cryopreservation does adversely impact angiogenesis and implantation in this setting. Although we have optimized freeze/thaw and autotransplantation procedures for use in several experiments [6,16,28,29], not all ovaries so-treated regained their function, and a certain portion became atretic [30]. This suggests that additional improvements in methodology are in order to enhance ovarian tissue survival.
Because similar levels of angiogenic factors were maintained after autotransplantation with or without prior cryopreservation, we suspect that angiogenesis (as mediated by VEGF and angpt-2) in transplanted states commonly proceeds this way. Likewise, the technique used for cryopreservation, whether vitrification or slow freezing, did not influence expression of angiogenic factors in heterotopic autotransplants. Hence, vitrification is validated as a useful method of ovarian tissue cryopreservation for autotransplantation.
In conclusion, autotransplantation of whole, cryopreserved ovaries beneath the abdominal walls of mice is feasible. Any loss of angiogenic factor expression due to cryopreservation is not method-dependent and is recovered after autotransplantation, similar to non-cryopreserved counterparts. Additional studies are needed to further characterize the roles of angiogenic factors under these circumstances.
Acknowledgments
This work was supported by the Clinical Research Fund (GNUH-CRF-2010-006), Gyeongsang National University Hospital.
References
1. Blatt J. Pregnancy outcome in long-term survivors of childhood cancer. Med Pediatr Oncol 1999;33:29-33. PMID: 10401494.


2. Aubard Y, Poirot C, Piver P, Galinat S, Teissier MP. Are there indications for ovarian tissue cryopreservation? Fertil Steril 2001;76:414-415. PMID: 11476802.


3. Oktay K, Economos K, Kan M, Rucinski J, Veeck L, Rosenwaks Z. Endocrine function and oocyte retrieval after autologous transplantation of ovarian cortical strips to the forearm. JAMA 2001;286:1490-1493. PMID: 11572742.


4. Oktay K. Successful human ovarian autotransplantation to the upper arm. Cancer 2005;103:1982-1983. PMID: 15786418.


5. Kim SS, Hwang IT, Lee HC. Heterotopic autotransplantation of cryobanked human ovarian tissue as a strategy to restore ovarian function. Fertil Steril 2004;82:930-932. PMID: 15482772.


6. Bedaiwy MA, Jeremias E, Gurunluoglu R, Hussein MR, Siemianow M, Biscotti C, et al. Restoration of ovarian function after autotransplantation of intact frozen-thawed sheep ovaries with microvascular anastomosis. Fertil Steril 2003;79:594-602. PMID: 12620446.


7. Oktay K, Buyuk E, Veeck L, Zaninovic N, Xu K, Takeuchi T, et al. Embryo development after heterotopic transplantation of cryopreserved ovarian tissue. Lancet 2004;363:837-840. PMID: 15031026.


8. Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J. Vascular-specific growth factors and blood vessel formation. Nature 2000;407:242-248. PMID: 11001067.


9. Hazzard TM, Molskness TA, Chaffin CL, Stouffer RL. Vascular endothelial growth factor (VEGF) and angiopoietin regulation by gonadotrophin and steroids in macaque granulosa cells during the peri-ovulatory interval. Mol Hum Reprod 1999;5:1115-1121. PMID: 10587365.


10. Gale NW, Yancopoulos GD. Growth factors acting via endothelial cell-specific receptor tyrosine kinases: VEGFs, angiopoietins, and ephrins in vascular development. Genes Dev 1999;13:1055-1066. PMID: 10323857.


11. Suri C, Jones PF, Patan S, Bartunkova S, Maisonpierre PC, Davis S, et al. Requisite role of angiopoietin-1, a ligand for the TIE2 receptor, during embryonic angiogenesis. Cell 1996;87:1171-1180. PMID: 8980224.


12. Thurston G, Rudge JS, Ioffe E, Zhou H, Ross L, Croll SD, et al. Angiopoietin-1 protects the adult vasculature against plasma leakage. Nat Med 2000;6:460-463. PMID: 10742156.


13. Holash J, Wiegand SJ, Yancopoulos GD. New model of tumor angiogenesis: dynamic balance between vessel regression and growth mediated by angiopoietins and VEGF. Oncogene 1999;18:5356-5362. PMID: 10498889.


14. Asahara T, Chen D, Takahashi T, Fujikawa K, Kearney M, Magner M, et al. Tie2 receptor ligands, angiopoietin-1 and angiopoietin-2, modulate VEGF-induced postnatal neovascularization. Circ Res 1998;83:233-240. PMID: 9710115.


15. Nishigaki A, Okada H, Tsuzuki T, Cho H, Yasuda K, Kanzaki H. Angiopoietin 1 and angiopoietin 2 in follicular fluid of women undergoing a long protocol. Fertil Steril 2011;96:1378-1383. PMID: 22000910.


16. Choi WJ, Lee JH, Park MH, Choi IY, Park JK, Shin JK, et al. Influence of the vitrification solution on the angiogenic factors in vitrificated mouse ovarian tissue. Obstet Gynecol Sci 2013;56:382-388. PMID: 24396817.



17. Maltaris T, Beckmann MW, Dittrich R. Fertility preservation for young female cancer patients. In Vivo 2009;23:123-130. PMID: 19368136.

18. Silber SJ. Ovary cryopreservation and transplantation for fertility preservation. Mol Hum Reprod 2012;18:59-67. PMID: 22205727.


19. Baird DT, Webb R, Campbell BK, Harkness LM, Gosden RG. Long-term ovarian function in sheep after ovariectomy and transplantation of autografts stored at -196 C. Endocrinology 1999;140:462-471. PMID: 9886858.


20. Abir R, Orvieto R, Raanani H, Feldberg D, Nitke S, Fisch B. Parameters affecting successful transplantation of frozenthawed human fetal ovaries into immunodeficient mice. Fertil Steril 2003;80:421-428. PMID: 12909508.


21. Israely T, Dafni H, Nevo N, Tsafriri A, Neeman M. Angiogenesis in ectopic ovarian xenotransplantation: multiparameter characterization of the neovasculature by dynamic contrastenhanced MRI. Magn Reson Med 2004;52:741-750. PMID: 15389965.


22. Kim SS, Yang HW, Kang HG, Lee HH, Lee HC, Ko DS, et al. Quantitative assessment of ischemic tissue damage in ovarian cortical tissue with or without antioxidant (ascorbic acid) treatment. Fertil Steril 2004;82:679-685. PMID: 15374714.


23. Ferrara N. Vascular endothelial growth factor: molecular and biological aspects. Curr Top Microbiol Immunol 1999;237:1-30. PMID: 9893343.


24. Friedman O, Orvieto R, Fisch B, Felz C, Freud E, Ben-Haroush A, et al. Possible improvements in human ovarian grafting by various host and graft treatments. Hum Reprod 2012;27:474-482. PMID: 22114111.


25. Isachenko V, Isachenko E, Reinsberg J, Montag M, van der Ven K, Dorn C, et al. Cryopreservation of human ovarian tissue: comparison of rapid and conventional freezing. Cryobiology 2007;55:261-268. PMID: 17931616.


26. Santos RR, Tharasanit T, Van Haeften T, Figueiredo JR, Silva JR, Van den Hurk R. Vitrification of goat preantral follicles enclosed in ovarian tissue by using conventional and solid-surface vitrification methods. Cell Tissue Res 2007;327:167-176. PMID: 16937112.


27. Dissen GA, Lara HE, Fahrenbach WH, Costa ME, Ojeda SR. Immature rat ovaries become revascularized rapidly after autotransplantation and show a gonadotropin-dependent increase in angiogenic factor gene expression. Endocrinology 1994;134:1146-1154. PMID: 8119153.


28. Bedaiwy MA, Falcone T. Harvesting and autotransplantation of vascularized ovarian grafts: approaches and techniques. Reprod Biomed Online 2007;14:360-371. PMID: 17359593.


29. Jeremias E, Bedaiwy MA, Gurunluoglu R, Biscotti CV, Siemionow M, Falcone T. Heterotopic autotransplantation of the ovary with microvascular anastomosis: a novel surgical technique. Fertil Steril 2002;77:1278-1282. PMID: 12057741.


30. Grazul-Bilska AT, Banerjee J, Yazici I, Borowczyk E, Bilski JJ, Sharma RK, et al. Morphology and function of cryopreserved whole ovine ovaries after heterotopic autotransplantation. Reprod Biol Endocrinol 2008;6:16PMID: 18402709.



Fig. 1
Flow diagram of experimental design: After 1 week of cryopreservation (vitrification or slow freezing), one of the paired mouse ovaries was analyzed for angiogenic factor expression; the other ovary was autotransplanted beneath the abdominal wall. After a 2-week autotransplantation period, angiogenic factors were assayed.
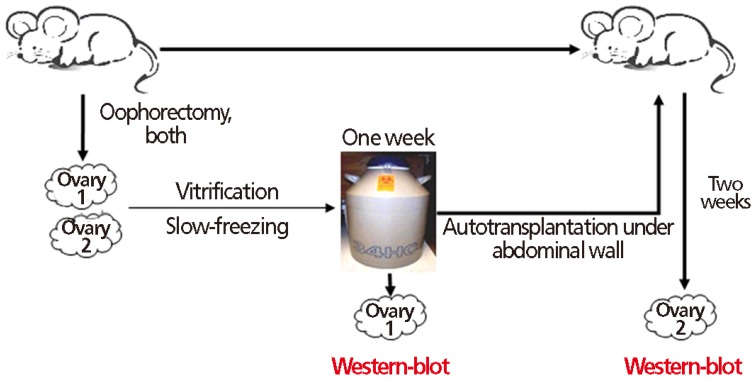
Fig. 2
Autotransplanted mouse ovary: (A) beneath the abdominal wall (arrow), to be assayed after 2 weeks, and (B) on dish with media.
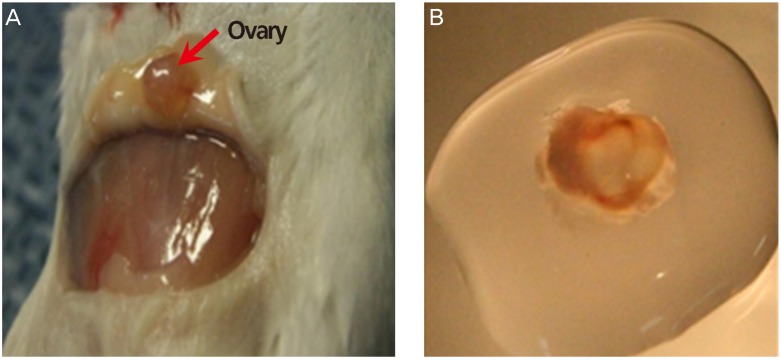
Fig. 3
Western blot analysis: quantitative analysis by group of (A) vascular endothelial growth factor (VEGF) and (B) angiopoietin-2 (angpt-2) after 1 week of vitrification or slow freezing (no cryopreservation of controls). β-actin was used as internal standard. Data were expressed as mean±standard error of three independent experiments. *P<0.05 vs. cryopreservation groups (vitrification and slow freezing).
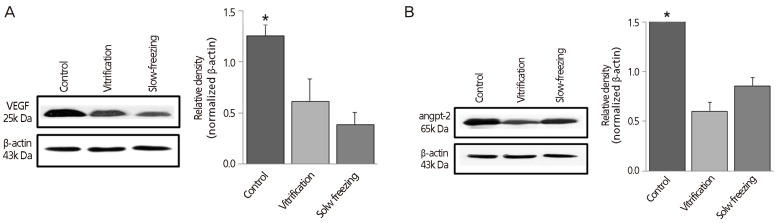
Fig. 4
Western blot analysis: quantitative analysis by group of (A) vascular endothelial growth factor (VEGF) and (B) angiopoietin-2 (angpt-2) after 2 weeks of autotransplantation beneath the abdominal wall (no cryopreservation of controls). β-actin was used as internal standard. Data expressed as mean±standard error of three independent experiments.
-
METRICS

-
- 2 Crossref
- 3,059 View
- 26 Download
- Related articles in Obstet Gynecol Sci
-
The expression of Thymosin beta4 with angiogenic factors in epithelial ovarian cancer.2008 May;51(5)



















