|
 |
- Search
| Obstet Gynecol Sci > Volume 57(4); 2014 > Article |
Abstract
Objective
To evaluate the effect of progesterone supplementation during the luteal phase on pregnancy outcome in natural frozen-thawed embyo transfer (FTET) cycles.
Methods
In this retrospective cohort study, 228 consecutive patients who underwent FTET cycles between January 2009 and September 2012 were included. One hundred forty-five patients received luteal progesterone support (P group) but 83 patients did not receive any progesterone supplementation during luteal phase (control group).
Results
There were no differences in patients' characteristics between the two groups. The two groups were similar with respect to the characteristics of previous fresh in vitro fertilization cycle in which embryos were cryopreserved including the numbers of oocytes retrieved, mature oocytes, fertilized oocytes, grade 1 or 2 embryos and frozen embryos. Also, significant differences were not observed between the P and control groups in clinical pregnancy rate, embryo implantation rate and multiple pregnancy rate. However, miscarriage rate was significantly lower in the P group and live birth rate was significantly higher in the P group than in the control group (P<0.05, P<0.05).
Controlled ovarian stimulation (COS) commonly results in the generation of supernumerary embryos, and therefore embryo cryopreservation and subsequent frozen-thawed embyo transfer (FTET) became popular and essential part of in vitro fertilization (IVF) and embryo transfer (ET) program. Frozen-thawed embyos should be replaced during the window of endometrial receptivity. Various protocols for replacement of frozen-thawed embryos in the optimum time have been developed. FTET can be performed during a natural ovulatory cycle, hormone replacement cycle or ovulation induction cycle [1]. A recent Cochrane review could not draw any conclusions on the superiority of any one regimen with respect to ongoing pregnancy rate [1]. Nevertheless, nowadays natural FTET cycle is becoming a common approach and favored option for patients with normal ovulatory cycles, because this natural cycle protocol does not require medication including exogenous estrogen and the time taken to complete FTET cycle is short.
Progesterone is a prerequisite for endometrial development and embryo implantation. Adequate progesterone exposure in addition to preceding estrogen priming is essential for transformation of the endometrium to a receptive phase [2]. In fertility proven women, endogenous production of progesterone from the corpus luteum is sufficient to support the embryo implantation in a natural ovulatory cycle. However, the women undergoing FTET are often subfertile or infertile, and their corpus luteal progesterone production may be insufficient during their natural cycles. Inadequate progesterone production during the luteal phase or early pregnancy period can result in implantation failure or spontaneous abortion [3,4]. Therefore, luteal phase progesterone supplementation may be needed if frozen embryo is transferred during a natural cycle to an infertile woman. So far, there are limited data with inconsistent results on luteal phase progesterone supplementation after FTET in natural cycles. Moreover, it is hard to find the study on the effect of luteal progesterone supplementation in natural FTET cycles in Korean infertile women.
Therefore, we performed a retrospective cohort study to investigate if vaginal progesterone supplementation during the luteal phase and early pregnancy period would improve the pregnancy outcome in infertile women who underwent natural FTET cycle.
This retrospective cohort study included 228 consecutive patients who underwent FTET in their natural ovulatory cycles between January 2009 and September 2012. One hundred forty-five patients received vaginal progesterone supplementation during the luteal phase in their natural FTET cycles (P group). In contrast, 83 patients did not receive any progesterone supplementation during luteal phase (control group). Only the first natural FTET cycle per patient was included in the present study. The institutional review board of the University of Ulsan College of Medicine, Asan Medical Center, approved the study.
All women were between 20 and 41 years of age, and they had regular ovulatory cycles of 24 to 35 days in length and body mass index (BMI) between 18 and 25 kg/m2. Patients were excluded from this study if they were found to have any endocrine and metabolic disorders such as diabetes mellitus and any significant pelvic pathology such as hydrosalpinx, uterine anomaly, or fibroids with uterine cavity distortion. Subjects who had any abnormalities that would interfere with adequate stimulation or a history of previous (within 12 months) or current abuse of alcohol or drugs were also excluded.
All women had previously undergone either conventional IVF or intracytoplasmic sperm injection (ICSI) with embryo cryopreservation. In all patients, cycle monitoring was started on cycle day 7 or 8 by transvaginal ultrasonography (ProSound SSD-5500, ALOKA, Tokyo, Japan) and repeated according to the size of the dominant follicle. When a mean diameter of the lead follicle reached 18 mm, 250 µg recombinant hCG (rhCG; Ovidrel, Merck Serono SA, Geneva, Switzerland) was administered subcutaneously to induce follicular maturation. On the day of ET, ovulation was confirmed by transvaginal ultrasonography. Cryopreserved zygotes at the 2 pronuclear (PN) stage were thawed on the 3rd day after hCG injection and cultured. We excluded the FTET cycles in which cryopreserved embryos at the cleaved or blastocyst stage were thawed and transferred from this study, in order to exclude the possibility of impact on the pregnancy outcome according to the different developmental stages of embryos frozen and freezing method.
ET was performed 5 days after hCG injection. On the day of ET, transvaginal color Doppler analysis was performed to assess the resistance index (RI) of the uterine arteries. One to four embryos were transferred into the uterus and both the embryo stage and grade were recorded for each transferred embryo. In the P group, luteal support was provided by administering 90mg of vaginal progesterone gel (Crinone gel 8%, Merck Serono SA, Geneva, Switzerland) once daily from the 2nd day after hCG injection and continued at least up to the day of serum β-hCG measurement for pregnancy test. If pregnancy was achieved, administration of vaginal progesterone gel at a same dose was continued until eleven to twelve weeks of gestation. Patients who served as controls did not receive any progesterone supplementation during luteal phase and early pregnancy period.
The β-hCG serum levels were measured by radioimmunoassay using a hCG MAIA clone kit (Serono Diagnostics, Woking, UK) with interassay and intraassay variances of <10% and 5%, respectively, 11 days after ET. Clinical pregnancy was defined as the presence of a gestational sac by ultrasonography, while miscarriage rate per clinical pregnancy was defined as the proportion of patients who failed to continue development before 20 weeks of gestation in all clinical pregnancies.
Mean values were expressed as mean±standard deviation. Student's t-test was used to compare mean values. Chi-square test and Fisher's exact test were used to compare fraction. Statistical significance was defined as P<0.05. All analyses were performed by using PASW ver. 18.0 (SPSS Inc. Chicago, IL, USA).
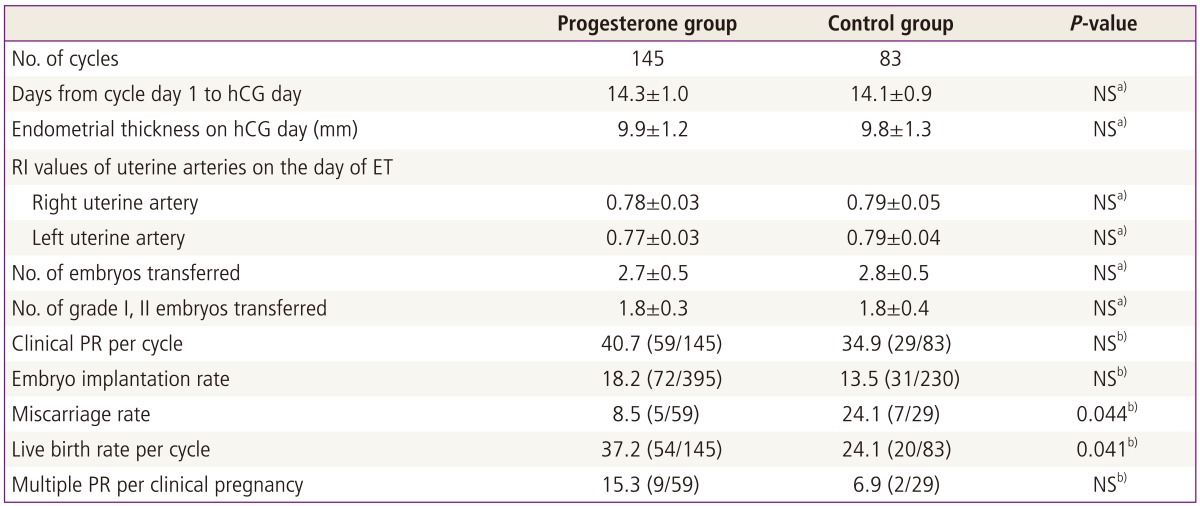
The two groups were comparable with respect to the ages of the patients and their spouses, duration of infertility, BMI, the proportion of nullipara, antral follicle count, antimüllerian hormone level and IVF/ICSI indications (Table 1).
Table 2 presents the comparison of characteristics of previous IVF/ICSI cycles in which 2PN zygotes were cryopreserved between the P and control groups. The two groups were similar in previous COS protocols and fertilization methods. There were no significant differences between the two groups with respect to the days of recombinant human follicle stimulating hormone (rhFSH), total dose of rhFSH administered, numbers of oocytes retrieved, mature oocytes, fertilized oocytes, grade I or II embryos, 2PN zygotes cryopreserved and blastocysts vitrified and the incidence of severe ovarian hyperstimulation syndrome (Table 2).
When the characteristics of FTET cycles were compared between the two groups, there were no significant differences in days from cycle day 1 to the day of hCG injection, endometrial thickness on the day of hCG injection, RI values of uterine arteries on the day of ET, and the numbers of total embryos transferred and grade I or II embryos transferred (Table 3). Although there were trends toward the increased clinical pregnancy rate and the increased embryo implantation rate with vaginal progesterone supplementation, the differences did not achieve the statistical significance. However, miscarriage rate was significantly lower and live birth rate was significantly higher in the P group than in the control group (P=0.044, P=0.041, respectively) (Table 3).
Progesterone is essential for the embryo implantation on the endometrium. The endometrium acquires receptivity to embryo implantation by adequate progesterone exposure in addition to preceding estrogen priming [2]. Estrogen in the follicular phase induces endometrial proliferation and the induction of progesterone receptors in the endometrium. Low number of endometrial progesterone receptors or low levels of circulating progesterone during the luteal phase may result in embryo implantation failure or miscarriage [2,4].
Endometrial receptivity can be compromised by ovarian stimulation [5]. The elevation of serum estradol to supraphysiologic levels by COS was prone to alter endometrial receptivity by causing an imbalance of estradiol/progesterone ratio [6] and suppressing luteinizing hormone (LH) production concentration after COS [7]. These conditions frequently results in luteal phase defect (LPD). Therefore, luteal phase progesterone supplementation after COS is important for successful embryo implantation and has been current practice in COS cycles.
Contrary to the COS cycles, endogenous progesterone concentration during the luteal phase is generally sufficient to support implantation in a natural ovulatory cycle. However, infertile women undergoing FTET in natural ovulatory cycles can be different and they may have suboptimal levels of endogenous progesterone after ovulation in their natural cycles. In addition, hCG administered for triggering oocyte maturation may result in LPD. hCG administered in stimulated IVF cycles would cause LPD by suppressing LH production through a short-loop feedback mechanism [8], although Tavaniotou and Devroey reported that hCG injection did not down-regulate LH secretion in the luteal phase of natural cycles in women with normal ovulation [9]. HCG results in the activation of adenylate cyclase to increase the intracellular levels of cyclic AMP and increased cyclic AMP induce LH receptor mRNA binding protein (LRBP) synthesis and/or activation presumably through stimulation of protein kinase A. These events promote increased LRBP binding to LH/hCG receptor mRNA, thereby down-regulating LH/hCG receptor expression [10].
In all patients included in the present study, hCG was administered for final oocyte maturation. Our hypothesis in this study was that luteal phase progesterone supplementation has a beneficial effect on pregnancy outcome after FTET in hCG-administered natural cycles. Actually, our results supported our hypothesis. Progesterone supplementation significantly reduced miscarriage rate and increased live birth rate in hCG-administered natural FTET cycles, although significant differences in clinical pregnancy rate and embryo implantation rate were not observed between progesterone supplementation group and no supplementation group. Our results suggest that hCG-administered natural FTET cycles may be associated with LPD, and LPD may be an etiologic factor for miscarriage after embryo implantation rather than a cause for implantation failure. Also, in the prospective randomized controlled trial by Bjuresten et al. [11], progesterone supplementation significantly increased the live birth rate after FTET in natural cycles. In their study, clinical abortion rate was also lower in the patient group receiving micronized vaginal progesterone at a dose of 400 mg twice a day from the ET day, but the difference did not achieve the statistical significance.
Contrary to the results of our study, Kyrou et al. [12] reported that luteal micronized vaginal progesterone supplementation at a dose of 200 mg three times a day from the day after hCG administration had no beneficial effect on ongoing pregnancy rate in hCG-induced natural FTET cycles. In their retrospective study, ongoing pregnancy rates in progesterone supplementation group and no progesterone supplementation group were just 21% and 22%, respectively, although embryo implantation rates in progesterone supplementation group and no progesterone supplementation group were 22.6% and 24.4%, respectively. These ongoing pregnancy rates in their study appears to be much lower than live birth rates in our present study. Their results may result from the small numbers of embryos transferred (in progesterone supplementation group and no supplementation group, 1.6 and 1.5, respectively).
In conclusion, our results suggest that vaginal progesterone supplementation for luteal support in hCG-administered natural FTET cycles is beneficial to the pregnancy outcome. The major disadvantage of the present study is its retrospective design and therefore larger prospective randomized trial with standardized methods is needed for the confirmation of our results.
References
1. Ghobara T, Vandekerckhove P. Cycle regimens for frozen-thawed embryo transfer. Cochrane Database Syst Rev 2008;(1):CD003414PMID: 18254019.


2. Critchley HO, Saunders PT. Hormone receptor dynamics in a receptive human endometrium. Reprod Sci 2009;16:191-199. PMID: 19208787.


3. Daya S. Luteal support: progestogens for pregnancy protection. Maturitas 2009;65(Suppl 1):S29-S34. PMID: 19913373.


4. Csapo AI, Pulkkinen MO, Ruttner B, Sauvage JP, Wiest WG. The significance of the human corpus luteum in pregnancy maintenance. I. Preliminary studies. Am J Obstet Gynecol 1972;112:1061-1067. PMID: 5017636.


5. Haouzi D, Assou S, Mahmoud K, Tondeur S, Reme T, Hedon B, et al. Gene expression profile of human endometrial receptivity: comparison between natural and stimulated cycles for the same patients. Hum Reprod 2009;24:1436-1445. PMID: 19246470.



6. Sharara FI, McClamrock HD. Ratio of oestradiol concentration on the day of human chorionic gonadotrophin administration to mid-luteal oestradiol concentration is predictive of in-vitro fertilization outcome. Hum Reprod 1999;14:2777-2782. PMID: 10548621.


7. Tavaniotou A, Albano C, Smitz J, Devroey P. Comparison of LH concentrations in the early and mid-luteal phase in IVF cycles after treatment with HMG alone or in association with the GnRH antagonist Cetrorelix. Hum Reprod 2001;16:663-667. PMID: 11278214.


8. Miyake A, Aono T, Kinugasa T, Tanizawa O, Kurachi K. Suppression of serum levels of luteinizing hormone by short- and long-loop negative feedback in ovariectomized women. J Endocrinol 1979;80:353-356. PMID: 220373.


9. Tavaniotou A, Devroey P. Effect of human chorionic gonadotropin on luteal luteinizing hormone concentrations in natural cycles. Fertil Steril 2003;80:654-655. PMID: 12969719.


10. Menon KM, Nair AK, Wang L. A novel post-transcriptional mechanism of regulation of luteinizing hormone receptor expression by an RNA binding protein from the ovary. Mol Cell Endocrinol 2006;246:135-141. PMID: 16406262.


11. Bjuresten K, Landgren BM, Hovatta O, Stavreus-Evers A. Luteal phase progesterone increases live birth rate after frozen embryo transfer. Fertil Steril 2011;95:534-537. PMID: 20579989.


12. Kyrou D, Fatemi HM, Popovic-Todorovic B, Van den Abbeel E, Camus M, Devroey P. Vaginal progesterone supplementation has no effect on ongoing pregnancy rate in hCG-induced natural frozen-thawed embryo transfer cycles. Eur J Obstet Gynecol Reprod Biol 2010;150:175-179. PMID: 20219279.


Table 2
Characteristics of previous fresh IVF cycles in which embryos were cryopreserved
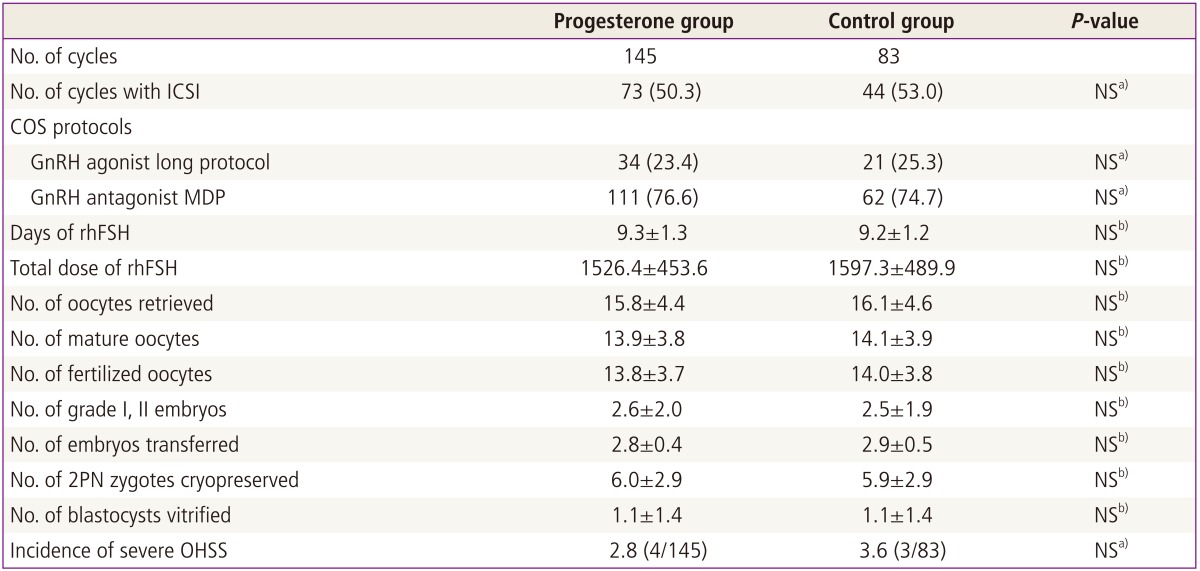
Values are mean±standard deviation or number (%).
IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection; NS, not significant; COS, controlled ovarian stimulation; MDP, multiple dose protocol; rhFSH, recombinant human follicle stimulating hormone; PN, pronuclear; OHSS, ovarian hyperstimulation syndrome.
a)Chi-square test or Fisher's exact test; b)Student's t-test.
-
METRICS

-
- 46 Crossref
- 4,326 View
- 73 Download
- Related articles in Obstet Gynecol Sci